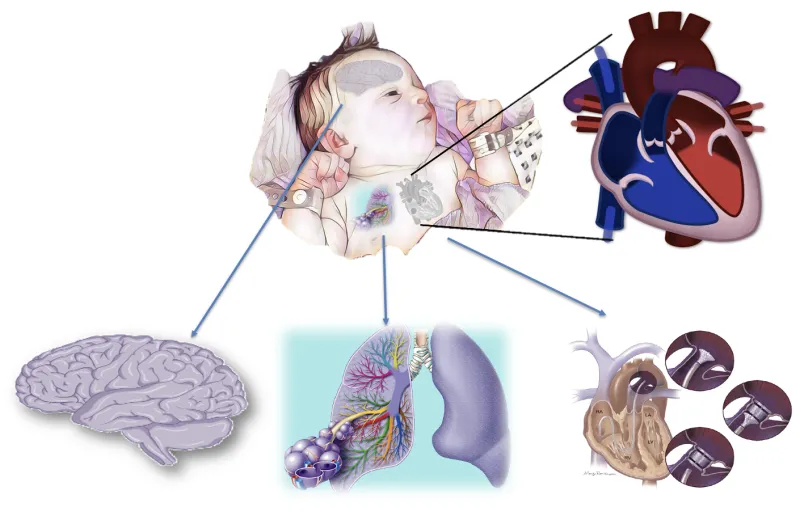
Heart disease
Acquired heart disease
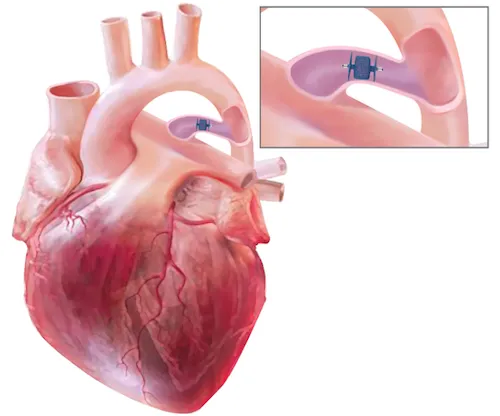
In 2019, the Food and Drug Administration approved a new transcatheter device to close PDAs in extremely-low birth weight newborns, making it possible for many more patients to benefit from this minimally invasive procedure. We established a Premature PDA Closure Program at Boston Children's Hospital to support neonates eligible for transcatheter device closure of the PDA, and we manage a consultation service for the New England region (16 referral centers) that facilitates transfer, care, and follow up for all babies who come to Boston Children's Hospital for device closure. The program has its own database, and its impact serves as resources for comparative reports and quality improvement initiatives; our data from over 300 consultations, 185 closures, original clinical manuscripts, and reviews has fueled two randomized control trials: (1) Percutaneous Intervention Versus Observational Trial of Arterial Ductus in Low weight Infants (PIVOTAL, NCT05547165);and (2) Milrinone for Prevention of Post-ligation Cardiac Syndrome Trial (MIDAS, NCT06679855).
The impact of definitive closure on cardiopulmonary mechanics and options to minimize the short- and long- term impact is also limited. Furthermore, decisions regarding optimal timing for device occlusion and their hemodynamic impact to minimize consequences of a persistent ductus arteriosus remain to be fully elucidated. Our group is partnering with regional and national organization with a focus on addressing crucial gaps to enable widespread use of algorithms and rigorously inform clinical decision-making, with the ultimate goal of improving neonatal outcomes in preterm infants with a hsPDA.